|
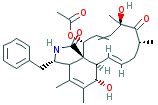
CYTOCHALASIN C |
| Synonyms. Cytochalasin C; (3S-(3R*,3aS*,6R*,6aS*,7E,10R*,12S*,13E,15S*,15aS*))-15-(Acetyloxy)-3,3a,6,6a, 9,10,12,15- octahydro- 6,12-dihydroxy-4,5,10,12-tetramethyl-3-(phenylmethyl)-1H-cycloundec(d)isoindole- 1,11(2H)-dione; 7(S),18-Dihydroxy-16(S),18(R)-dimethyl-10-phenyl(11)cytochalasa-5,13(E),19(E)-triene-1,17-dione 21(R)-acetate; |
|
|
| PRODUCT IDENTIFICATION | |
|
CAS RN |
22144-76-9 |
|
EINECS RN |
244-803-6 |
|
FORMULA |
C30H37NO6 |
|
MOLE WEIGHT |
507.62 |
|
H.S CODE |
2933.79.0800 |
|
SMILES |
N1[C@@H]([C@@H]2C(=C([C@H]([C@H]3[C@]2(C1=O)[C@@H](C=C[ C@@] (C([C@H](CC=C3)C) =O)(C)O)OC(=O)C)O)C)C)Cc1ccccc1 |
|
CLASSIFICATION |
Mycotoxin, Actin inhibitor |
|
EXTRA NOTES |
|
|
|
| PHYSICAL AND CHEMICAL PROPERTIES | |
|
PHYSICAL STATE. |
white powder |
|
MELTING POINT |
2 ~ C |
|
BOILING POINT |
2 ~ C |
|
DENSITY |
|
|
SOLUBILITY IN WATER |
|
| SOLVENT SOLUBILITY |
|
|
VAPOR DENSITY |
|
|
log P(octanol-water) |
|
|
VAPOR PRESSURE |
|
|
AUTOIGNITION TEMP |
|
| pK |
|
|
REFRACTIVE INDEX |
|
|
FLASH POINT |
|
|
|
| STABILITY AND REACTIVITY | |
| STABILITY | Stable under normal conditions. |
|
INCOMPATIBLE MATERIALS |
Strong oxidizing agents |
| POLYMERIZATION |
Has not been reported |
|
NFPA RATINGS |
Health: 1, Flammability: 0, Reactivity: 0 |
|
|
| EXTERNAL LINKS & GENERAL DESCRIPTION |
|
USA.gov - Cytochalasin C Wikipedia Linking - Cytochalasin Google Scholar Search - Cytochalasin C U.S. National Library of Medicine - Cytochalasin C PubChem Compound Summary - Cytochalasin C KEGG (Kyoto Encyclopedia of Genes and Genomes) - Cytochalasin C NCBI (http://www.ncbi.nlm.nih.gov/) - Cytochalasin C Cytochalasin A is a metabolite of the fungus Drechslera (previously Heiminthosporium) dematioideum. Cytochalasin A is sulfhydryl-reactive, and was shown to inhibit growth and sugar uptake in a Saccharomyces strain. Unlike Cytochalasin B, Cytochalasin C and Cytochalasin D are isomeric metabolites of Metarrhizium anisopliae. Cytochalasin D possesses antibiotic and antitumor activity. It also impairs maintenance of long term potentiation (LTP) of actin filaments. It is implicated in promoting conditions favorable for depolymerizing actin. sigmaaldrich |
|
|
| SALES SPECIFICATION | |
|
APPEARANCE |
white powder |
|
ASSAY |
98% min |
|
|
| TRANSPORT & REGULATORY INFORMATION | |
|
UN NO. |
1544 |
| HAZARD CLASS |
6.1 |
| PACKING GROUP | II |
|
|
| SAFETY INFORMATION | |
|
HAZARD OVERVIEW |
GHS (Globally Harmonised System) Classification: Acute toxicity (Oral). Acute toxicity (Inhalation). Acute toxicity (Dermal). Reproductive toxicity. Hazard statements: Fatal if swallowed or in contact with skin. Fatal if inhaled. Suspected of damaging fertility or the unborn child. |
| SIGNAL WORD | Danger |
|
PICTOGRAMS |
|
|
HAZARD STATEMENTS |
H300 + H310-H330-H361 |
|
P STATEMENTS |
P260-P264-P280-P284-P302 + P350-P310 |
| EC DIRECTIVES |
|
| HAZARD CODES |
|
|
RISK PHRASES |
63-26/27/28 |
|
SAFETY PHRASES |
28-36/37-45 |
|
|
| PACKING |
|
Preserve in light-resistant and well-closed containers |
|
|